WHAT'S HOLDING PATIENTS BACK?
Administration challenges and dosing frequency associated with managing hemophilia A1,2


Bentlee lives with hemophilia A
Many patients prefer less frequent dosing...3,a
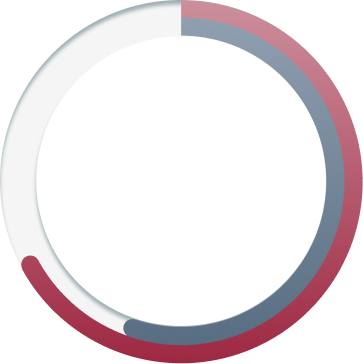
YET ONLY
12%
(N=6087)
USED AN EVERY-4-WEEK FVIII MIMETIC4,b
WHY AREN'T MORE PEOPLE FOLLOWING AN EVERY-4-WEEK DOSING REGIMEN DESPITE IT BEING AVAILABLE?
aBased on data from a cross-sectional, multicenter, observational study from 2018 to 2019 at hemophilia treatment centers in Korea (among 505 patients aged ≥16 years) with hemophilia A. Treatment satisfaction was measured using the treatment satisfaction questionnaire for medication (TSQM).3
bBased on qualitative survey data captured as of Q1 2025.4
ACCORDING TO World Federation of Hemophilia,
PATIENTS MAY TRADE
ADMINISTRATION BURDEN
FOR BLEED PROTECTION5
HIGH INJECTION VOLUME2,6
INJECTIONS AND
INFUSIONS CAN BE TOUGH
In a real world study of 354 patients with hemophilia A who were surveyed reported the following4,12
with FVIII mimetics (N=85) and FVIII prophylaxis (N=104), respectively, at least sometimes4,12
Response to survey questions: "Due to your treatment (while injecting or after), how often do you have [soreness, physical discomfort, pain, bruising, blown or ruptured veins, or problems due to scarring or scar tissue]?" Patients responded on a 5-point scale: not at all/never, a little/rarely, somewhat/sometimes, very/often, or extremely/always.1
Survey responses were collected in a cross-sectional study design. Responses in individuals over time may vary and may not account for continuous patient participation.
Data from an observational study including 354 patients with hemophilia A, conducted from 2020 to 2022, were collected through online surveys as part of the Hemo-TEM questionnaire with PicnicHealth. Patients responded to questions using a 5-point scale: never, rarely, sometimes, often, or always. Limitations may include selection bias due to the cross-sectional study design, variability in individual responses over time and does not account for continuous patient participation.
TREATMENTS MAY DISRUPT SOME PATIENTS' LIFESTYLES4,12
In a real-world observational study of 354 patients with hemophilia A4,12
16%
(N=85)
On FVIII
MIMETIC
26%
(N=104)
ON FVIII
PROPHYLAXIS
REPORTED CURRENT TREATMENT INTERFERED WITH...
TRAVEL AND VACATIONS
...at least sometimes4,12
SOME PATIENTS ALSO REPORTED THAT CURRENT TREATMENT INTERFERED WITH SOCIAL ACTIVITIES, DAILY ACTIVITIES, WORK, AND SCHOOL AT LEAST SOMETIMES.
Response to survey questions: "How much does taking your current treatment interfere with [your travel or vacations, social activities, daily activities, work, or school]?" Patients responded on a 5-point scale: not at all/never, a little/rarely, somewhat/sometimes, very/often, or extremely/always.4
Survey responses were collected in a cross-sectional study design. Responses in individuals over time may vary and may not account for continuous patient participation.4
Data from an observational study including 354 patients with hemophilia A, conducted from 2020 to 2022, were collected through online surveys as part of the Hemo-TEM questionnaire with PicnicHealth. Patients responded to questions using a 5- point scale: never, rarely, sometimes, often, or always. Limitations may include selection bias due to the cross-sectional study design, variability in individual responses over time and does not account for continuous patient participation.
Luke lives with Hemophilia A
Are patients
missing doses?
In a secondary analysis of data from a real-world study of 431 adults with hemophilia A4
55%
(N=44)
On FVIII
MIMETIC
67%
(N=257)
ON FVIII
PROPHYLAXIS
STATED THEY
postponed,
delayed or
missed a dose
AT LEAST SOMETIMES.1
Response to survey questions: Physicians were asked to indicate how adherent the patient is to their current hemophilia regimen on a 4-point scale: fully adherent, sub-optimally adherent, non-adherent, or not applicable. Patients were asked "On a scale from 1-10, where 1 is "Not at all" and 10 is "Fully", how adherent do you think you are to your hemophilia treatment regimen?"
Top patient-reported reasons FOR
non-adherence4
FVIII mimetic4
Forgetfulness
Difficulty following treatment regimen
Needle phobia
Interference with daily/social life
Difficulty attending appointments
FVIII prophylaxis4
Venous access issues
Side effects experienced
Forgetfulness
Difficulty attending appointments
Too frequent
Response to survey questions: Patients were asked to identify the reason or reasons for missed doses or being non-adherent; the most common are summarized here by treatment type.
Secondary analysis of data collected as part of a repeated cross-sectional, retrospective observational study of 431 adult patients with hemophilia A in the US. Data collected as part of the CHESS US data platform were used from 2019, 2023, and 2024. Information was collected using two questionnaire forms: a web-based 'case record form' (CRF), completed by physicians, and a 'patient and public involvement element' (PPIE) completed by patients. Limitations included the retrospective, non-interventional design and voluntary participation, which may introduce selection and recall bias.
Nikola lives with hemophilia A
References
- Brod M, Bushnell DM, Neergaard JS, Waldman LT, Busk AK. Understanding treatment burden in hemophilia: development and validation of the Hemophilia Treatment Experience Measure (Hemo-TEM). J Patient Rep Outcomes. 2023;7(1):17. Published 2023 Feb 23.
- Usach I, Martinez R, Festini T, Peris JE. Subcutaneous injection of drugs: literature review of factors influencing pain sensation at the injection site. Adv Ther. 2019;36(11):2986-2996
- Park YS, Hwang TJ, Cho GJ, et al. Patients' and parents' satisfaction with, and preference for, haemophilia A treatments: a cross-sectional, multicentre, observational study. Haemophilia. 2021;27(4):563-573.
- Data on file. Novo Nordisk Inc; Plainsboro, NJ.
- Srivastava A, Santagostino E, Dougall A, et al. WFH Guidelines for the Management of Hemophilia, 3rd edition [published correction appears in Haemophilia. 2021;27(4):699]. Haemophilia. 2020;26(suppl 6):1-158.
- Hemlibra® [package insert]. South San Francisco, CA: Genentech, Inc; 2024.
- Arendt-Nielsen L, Egekvist H, Bjerring P. Pain following controlled cutaneous insertion of needles with different diameters. Somatosens Mot Res. 2006;23(1-2):37-43.
- World Federation of Hemophilia. Protocols for the treatment of hemophilia and von Willebrand disease. WFH. April 2008. Accessed January 9, 2025. https://www1.wfh.org/publication/files/pdf-1137.pdf.
- Mannucci PM. Benefits and limitations of extended plasma half-life factor VIII products in hemophilia A. Expert Opin Investig Drugs. 2020;29(3):303-309.
- Wiley RE, Khoury CP, Snihur AWK, et al. From the voices of people with haemophilia A and their caregivers: challenges with current treatment, their impact on quality of life and desired improvements in future therapies. Haemophilia. 2019;25(3):433-440.
- Hakimi Z, Ghelani R, Bystrická L, et al. Patient Experience of Living With Hemophilia A: a conceptual model of humanistic and symptomatic experience in adolescents, adults, and children. J Health Econ Outcomes Res. 2024;11(2):95-102.
- Wheeler AP, Cibelli E, Hanson Get al. Treatment and disease burden in a cohort of people with haemophilia without inhibitors in the United States. Haemophilia. 2025;31(5):912-921.